(March 2026) – Early 2026 has seen the implementation of the final phase of the Carbon Border Adjustment Mechanism (CBAM), which extends the carbon pricing logic that is already central to the EU Emissions Trading System (EU ETS) to imported goods. This approach to managing emissions, particularly GHG (Greenhouse Gas) emissions, underscores the urgency for hard-to-abate sectors to identify new decarbonisation solutions. At the same time, solutions that valorise the industry’s own waste by repurposing it as raw material have become critically important.
While the ceramic industry has already made enormous efforts to reduce emissions and improve sustainability, innovative solutions for decarbonising the current production system and further improving the sector’s already high degree of circularity are becoming increasingly important.

This is the context behind the CCS4CER project: Carbon Capture Storage and CO2 Mineralisation for the Ceramic Industry, financed by the Emilia-Romagna Region’s European funds (PR-FESR 2021/2027, Axis 1 Research, Innovation and Competitiveness, Action 1.1.2) (www.ccs4cer.it/). Now in its third year, CCS4CER has delivered concrete results both in the study of carbon dioxide capture plant solutions and in the valorisation of the primary industrial waste currently generated by the ceramic sector and other energy-intensive industries: spent lime from bag filters used for the deacidification of flue gases originating from the kilns. Coordinated by Centro Ceramico, the project leverages the expertise of highly specialised laboratories within the Emilia-Romagna Region’s High Technology Network, including CIRI-FRAME (University of Bologna), LEAP (Milan Polytechnic’s Piacenza site) and Romagna Tech, alongside industrial partners such as Ascot Gruppo Ceramiche, Panariagroup Industrie Ceramiche S.p.A. and SACMI Cooperativa Meccanici Imola S.C.).
An initial sampling campaign of spent lime from various ceramic plants provided a complete chemical and physical characterisation. The capacity for CO2 sequestration using this waste is directly correlated to the quantity of unreacted Ca(OH)2 in the sample, which varies considerably according to the specific acid gas abatement protocols used by different companies. Samples analysed showed a high CO2 sequestration capacity, ranging between 190 and 390 grams of CO2 per kg of waste. The research explored CO2 mineralisation using wet (solid-liquid-gas) batch and semi-continuous processes across different scales.
During the study, various reaction conditions were explored by modifying operating pressures, flows and compositions of the CO2-containing gas, alongside suspension concentrations and waste types. The reaction profiles, established through continuous analysis of CO2 consumption, allowed for the study of process kinetics. The results demonstrate that the most influential factors on wet mineralisation efficiency are the liquid-solid ratio, CO2 flow and reaction time. Data also suggests that CO2 removal via this waste mineralisation is highly efficient even under mild conditions (low temperature and pressure) and with diluted CO2 concentrations.
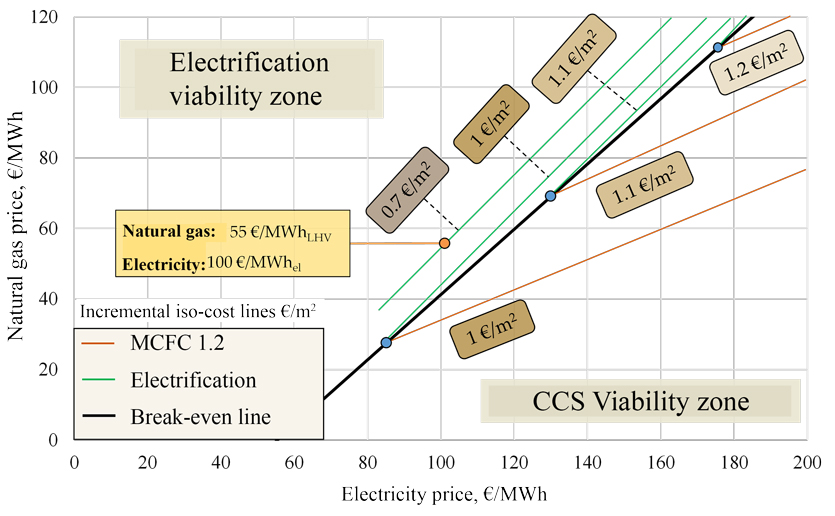
Incremental ceramic tile production cost trends relative to electricity (x-axis) and natural gas (y-axis) prices for MCFC (bottom right) and electrification (top left) technologies.
Testing carried out to date indicates that mineralisation reduces the release of pollutants from spent lime. In these tests, the material was reclassified from “hazardous special waste” to “non-hazardous waste”, offering significant benefits even for landfill disposal.
The study also investigated the reuse of mineralised spent lime in cementitious binders, a strategy that enables waste from one industrial sector to become a resource for another. The cement industry has long been in search of solutions to reduce clinker content and lower the overall environmental impact of its products. It has studied the use of mineralised material both as a filler to replace natural limestone (limestone Portland cements) and as a component of a new class of cements with reduced environmental impact containing limestone and calcined clay (LC3, Limestone Calcined Clay Cement). The test results show that the behaviour of the mineralised product in cement mortars is entirely comparable to pure limestone in terms of mechanical performance and chemical-physical characteristics and even offers benefits in terms of performance in LC3-based mortars. It has been confirmed that, in cement mortars, the release of any heavy metals present in the original spent lime into the aqueous phase is reduced.
To evaluate technologies for capturing CO2 from flue gases, simulations were conducted using industrial data from a plant producing 10,000 m2 of tiles per day (1 cm thick). Without capture systems, the plant consumes 10.3 MWPCI of natural gas and emits 18 kt/year of CO2. The production process is decarbonised using molten carbonate fuel cells (MCFCs), which allow for CO2 removal from flue gases and the production of decarbonised electricity to power the captured CO2 compression systems. Operating at around 600°C, MCFCs can be integrated into the production process to improve energy efficiency, albeit with increased plant complexity. Fuel cell performance was compared against amine-based chemical absorption and process electrification to replace natural gas combustion. Techno-economic analysis shows that while solvent-based solutions achieve 90% capture, they incur the highest decarbonisation costs among the cases analysed due to unfavourable scale factors. The MCFC system reduces CO2 by 84% and lowers natural gas consumption to 9.4 MWPCI. Electrification achieves 94% decarbonisation, emitting only the CO2 from the firing reactions, but requires significant plant modifications and uses 11.3 MWel. Both solutions increase ceramic tile production costs by €0.7-1.2/m2, representing a total production cost increase of between 8% and 22%, depending on product quality and energy prices.
Authors:
Riccardo Pascolo and Elisa Franzoni, Centro Ceramico, Sassuolo, Modena
Maria Chiara Bignozzi, Dipartimento di Ingegneria Civile, Chimica, Ambientale e dei Materiali, Bologna
Annamaria Catalano, Alessandro Allegri e Stefania Albonetti, CIRI-FRAME, University of Bologna, Dipartimento di Chimica Industriale, Bologna
Roberto Scaccabarozzi, Laboratorio Energia e Ambiente Piacenza (LEAP), Piacenza
Letizia Cretola, Politecnico di Milano, Dipartimento di Energia, Milano



 Architects
Architects
 Construction firms
Construction firms


